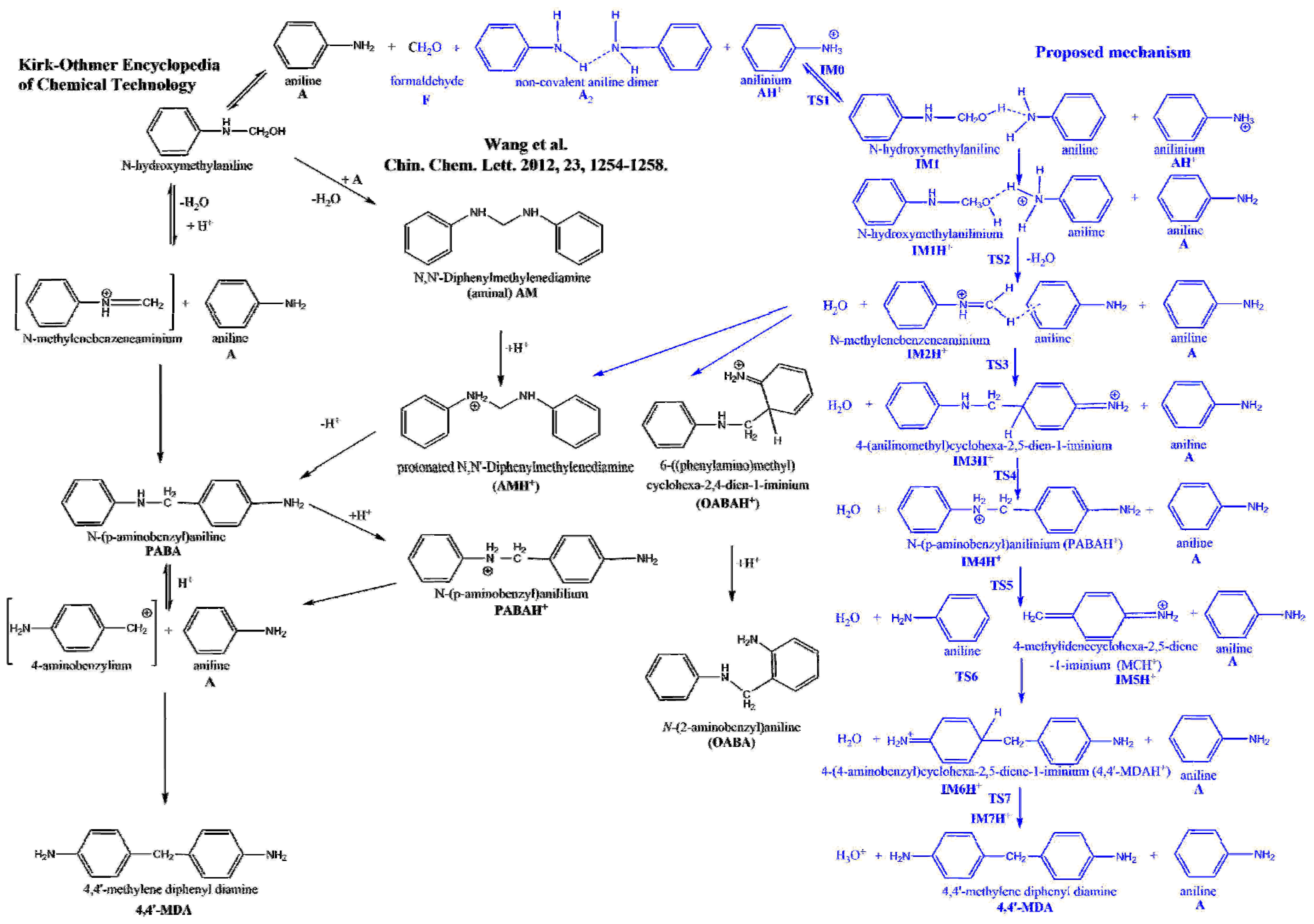
Chemical Name:CAS Number:Quantity Stored:Click here to enter textClick here to enter textClick here to enter textClick here to enter textClick here to enter textClick here to enter textSection 2 – Potential HazardsPerchloric acid. Perchloric acid is a strong mineral acid commonly used as a laboratory reagent. It is a clear, colorless liquid with no odor. Most perchloric acid is sold as solutions of 60% to 72% (w/w) acid in water. Perchloric acid is considered one of the strongest superacids. It is highly reactive with metals, dangerously corrosive, and readily forms explosive mixtures. In addition to being a corrosive liquid, while not combustible, under some circumstances perchloric acid may act as an oxidizer and/or present an explosion hazards.
Perchloric acid is non-combustible but may ignite other combustible materials and increase the intensity of a fire. Solutions of 85% (w/w) present a serious explosion hazard. They are unstable and can decompose explosively or spontaneously combust at ordinary temperatures if mixed with organic compounds. Heating produces toxic fumes of chlorine compounds.
Perchloric acid is a mineral acid with the formula HClO 4.Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid and nitric acid.It is a powerful oxidizer when hot, but aqueous solutions up to approximately 70% by weight at room temperature are generally safe, only showing strong acid features and no oxidizing properties. Polypropylene Chemical Compatibility Chart: Check the chemical compatibility of Polypropylene with various chemicals, solvents, alcohols and other products. Shop Polypropylene. Please Note: The information in this chart has been supplied by reputable sources and is to be used ONLY as a guide in selecting equipment for appropriate chemical compatibility.
Under some circumstances, perchloric acid vapors form perchlorates in ductwork, which are shock sensitive. Many heavy metal perchlorates and organic perchlorate salts are extremely shock sensitive explosives; the ammonium, alkali metal, and alkali earth perchlorates are somewhat less hazardous. Solutions of 72% w/w) are good oxidizers. Temperature increases the oxidizing power of perchloric acid, and hot concentrated solutions are very dangerous. Evaporation of a spill of the.
Storage & Use Location:Enter Building and Room Number(s)Storage Conditions:Enter Storage Conditions.(For example: room temperature, refrigerator, inert atmosphere, glove box. Acetic acidGlycol ethersAcetic anhydrideHydriodic acidAlcoholsHydrochloric acidAnilineHypophosphitesAntimony compounds (trivalent)KetonesBismuthLead oxide mixturesDehydrating agentsNitrogen triiodideDiethyl etherNitrosophenolFormaldehyde mixturesOrganic matter (e.g. Paper, wood, charcoal, rags, cotton, etc.)FluorineSodium iodideGlycerineSulfoxides and Sulfur trioxideGlycolsTransport Requirements:. Transportation of hazardous chemicals should follow the guidelines for chemicals as described in the BU. For bulk transport, chemicals should be transported using a clean cart. The materials themselves must be in sealed containers, clearly labeled with the contents name and applicable hazard classification(s).Section 8 – Spill and Accident ProceduresEmergency procedures for chemical exposure.
Remove any contaminated clothing as quickly as possible. Flush any exposed skin or hair with water for at least 15 minutes. Do not hesitate in activating a safety shower. For chemical exposure to the eye, flush with water for at least 15 minutes at an emergency eyewash station. Hold eyelids open while flushing. Seek appropriate medical assistance. If transported to hospital, bring a Safety Data Sheet (SDS) for the material you were exposed to, if able.Emergency procedures for chemical spill or accident.
Remove personnel from the immediate area. If the spill is small, dilute the perchloric acid with water and clean.
Do not attempt to handle a large spill/reaction/fire, or one in which you are not trained or equipped for. Turn off all ignition sources if this can be done safely, vacate the area, and call for assistance. Follow the reporting procedures on the Emergency Flip Chart.Section 9 – Waste DisposalPerchlorate contamination testSurfaces can also be tested to check for perchlorate contaminations:. Diphenylamine Test: Dissolve one gram of diphenylamine in 10 mL of “1 to 1” (18N) sulfuric acid to form a diphenylamine sulfate solution.
Using a medicine dropper, apply this solution to the test surface. The liquid turns black upon contact with perchlorate. The solution also reacts with nitrates, but turns blue. Methylene Blue Test: Use 0.4% solution of methylene blue in water. Add a few drops of indicator solution to about 25 mL of trial solution, such as water used to test rinse from a length of potentially contaminated duct.
Perchlorates will produce a violet precipitate.Waste disposal. Perchloric acid waste should never be mixed with other organic wastes. It should be kept in clearly identified clear glass containers labelled “Perchloric Acid Waste”.
If a bottle containing perchloric acid has turned dark and has crystals forming around the bottom of the bottle, there is a potential explosion hazard. Do not touch the container, as it might be highly unstable and shock sensitive. Any attempts to move or open the container might result into an explosion. Visually inspect the container in order to identify the content and to look for an expiration date.
All waste and contaminated disposables should be disposed of as hazardous waste, including labeling as hazardous waste and use of secondary containment. Charles River Campus waste disposal requests should be directed to EHS at. Medical Campus and BMC waste disposal requests should be directed toSection 10 – Lab Specific Changes.
.Acid strength refers to the tendency of an, symbolised by the HA, to dissociate into a, H +, and an, A −. The of a strong acid in solution is effectively complete, except in its most concentrated solutions.HA → H + + A −Examples of are (HCl), (HClO 4), (HNO 3) and (H 2SO 4).A weak acid is only partially dissociated, with both the undissociated acid and its dissociation products being present, in solution, in equilibrium with each other.HA ⇌ H + + A −.(CH 3COOH) is an example of a weak acid. The strength of a weak acid is quantified by its, p K a value.The strength of a weak acid may depend on substituent effects.
The strength of an acid is dependent on the for the atom to which the proton may be attached. Acid strength is solvent-dependent. For example, is a strong acid in aqueous solution, but is a weak acid when dissolved in. Contents.Measures of acid strength The usual measure of the strength of an acid is its ( K a), which can be by methods. Stronger acids have a larger K a and a smaller logarithmic constant (p K a = −log K a) than weaker acids. The stronger an acid is, the more easily it loses a proton, H +.
Two key factors that contribute to the ease of are the of the H—A bond and the size of atom A, which determine the strength of the H—A bond. Acid strengths also depend on the stability of the conjugate base.While the p K a value measures the tendency of an acidic solute to transfer a proton to a standard solvent (most commonly water or ), the tendency of an acidic solvent to transfer a proton to a reference solute (most commonly a weak base) is measured by its, the H 0 value.
Although these two concepts of acid strength often amount to the same general tendency of a substance to donate a proton, the p K a and H 0 values are measures of distinct properties and may occasionally diverge. For instance, hydrogen fluoride, whether dissolved in water (p K a = 3.2) or DMSO (p K a = 15), has p K a values indicating that it undergoes incomplete dissociation in these solvents, making it a weak acid. However, as the rigorously dried, neat acidic medium, hydrogen fluoride has an H 0 value of –15, making it a more strongly protonating medium than 100% sulfuric acid and thus, by definition, a. (To prevent ambiguity, in the rest of this article, 'strong acid' will, unless otherwise stated, refer to an acid that is strong as measured by its p K a value (p K a. Image of a strong acid mostly dissociating.
The small red circles represent H + ions.A strong acid is an acid that dissociates according to the reactionHA + S ⇌ SH + + A −where S represents a solvent molecule, such as a molecule of water or, to such an extent that the concentration of the undissociated species HA is too low to be measured. For practical purposes a strong acid can be said to be completely dissociated. An example of a strong acid isHCl → H + + Cl − (in aqueous solution)Any acid with a p K a value which is less than about -2 is classed as a strong acid. This results from the very high of solutions with a of 1 or less and is known as the.The following are strong acids in aqueous and dimethyl sulfoxide solution. The values of p K a, cannot be measured experimentally. The values in the following table are average values from as many as 8 different theoretical calculations.Estimated values AcidFormulain waterin DMSOHCl-5.9 ± 0.4−2.0 ± 0.6HBr-8.8 ± 0.8−6.8 ± 0.8HI-9.5 ± 1−10.9 ± 1HCF 3SO 3−14 ± 2−14 ± 2HClO 4−15 ± 2−15 ± 2Also, in water. HNO 3 p K a = -1.6.
H 2SO 4 (first dissociation only, p K a1 ≈ −3): (p. 171)The following can be used as protonators in.
HSbF 6. HFSO 3SbF 5. HCHB 11Cl 11. HFSO 3 (p K a = −6.4), such as (tosylic acid) are a class of strong organic. Some sulfonic acids can be isolated as solids. Functionalized into is an example of a substance that is a solid strong acid.Weak acids.
Main article:The experimental determination of a p K a value is commonly performed by means of a. A typical procedure would be as follows. A quantity of strong acid is added to a solution containing the acid or a salt of the acid, to the point where the compound is fully protonated. The solution is then titrated with a strong baseHA + OH − → A − + H 2Ountil only the deprotonated species, A −, remains in solution.
At each point in the titration pH is measured using a and a. The equilibrium constant is found by fitting calculated pH values to the observed values, using the method of.Conjugate acid/base pair It is sometimes stated that 'the conjugate of a weak acid is a strong base'. Such a statement is incorrect. For example, acetic acid is a weak acid which has a K a = 1.75 x 10 −5.
Its conjugate base is the ion with K b = 10 −14/ K a = 5.7 x 10 −10 (from the relationship K a × K b = 10 −14), which certainly does not correspond to a strong base. The conjugate of a weak acid is often a weak base and vice versa.Acids in non-aqueous solvents The strength of an acid varies from solvent to solvent. An acid which is strong in water may be weak in a less basic solvent, and an acid which is weak in water may be strong in a more basic solvent.
According to, the solvent S can accept a proton.HA + S ⇌ A − + HS +.For example hydrochloric acid is a weak acid in solution in pure, HO 2CCH 3, which is more acidic than water.HO 2CCH 3 + HCl ⇌ (HO) 2CCH 3 + + Cl −The extent of ionization of the decreases in the order HI HBr HCl. Acetic acid is said to be a for the three acids, while water is not.: (p.
217)An important example of a solvent which is more basic than water is, DMSO, (CH 3) 2SO. A compound which is a weak acid in water may become a strong acid in DMSO.
Is an example of such a substance. An extensive bibliography of p K a values in solution in DMSO and other solvents can be found at.are strong acids even in solvents of low dielectric constant. Examples of superacids are. Some superacids can be crystallised. They can also quantitatively stabilize. Factors determining acid strength The inductive effect In organic carboxylic acids, an electronegative substituent can pull electron density out of an acidic bond through the, resulting in a smaller p K a value.
The effect decreases, the further the electronegative element is from the carboxylate group, as illustrated by the following series of.StructureNamep K a2-chlorobutanoic acid2.863-chlorobutanoic acid4.04-chlorobutanoic acid4.5butanoic acid4.5Effect of oxidation state In a set of of an element, p K a values decrease with the oxidation state of the element. The oxoacids of chlorine illustrate this trend.: (p. 171) StructureNameOxidationstatep K a7-8 †5-132.017.53† theoreticalReferences. Liang, Joan-Nan Jack (1976).
Hamilton, Ontario: McMaster University. P. 94. Miessler G.L. And Tarr D.A. Inorganic Chemistry (2nd ed., Prentice-Hall 1998, p.170). Porterfield, William W.
Inorganic Chemistry (Addison-Wesley 1984) p.260. Trummal, Aleksander; Lipping, Lauri; Kaljurand, Ivari; Koppel, Ilmar A.; Leito, Ivo (2016). 'Acidity of strong acids in water and dimethyl sulfoxide'. 120: 3663–3669. Bell, R.
(1973), The Proton in Chemistry (2nd ed.), Ithaca, NY: Cornell University Press. ^ Housecroft, C. E.; Sharpe, A. Inorganic Chemistry (2nd ed.).
Prentice Hall. ^ Guthrie, J.P. 'Hydrolysis of esters of oxy acids: p K a values for strong acids'. 56 (17): 2342–2354. Martell, A.E.; Motekaitis, R.J. Determination and Use of Stability Constants.
Chapter 4: Experimental Procedure for Potentiometric Measurement of Metal Complex Equilibria. Zhang, Dingliang; Rettig, Stephen J.; Trotter, James; Aubke, Friedhelm (1996).
'Superacid Anions: Crystal and Molecular Structures of Oxonium Undecafluorodiantimonate(V), H 3OSb 2F 11, Cesium Fluorosulfate, CsSO 3F, Cesium Hydrogen Bis(fluorosulfate), CsH(SO 3F) 2, Cesium Tetrakis(fluorosulfato)aurate(III), CsAu(SO 3F) 4, Cesium Hexakis(fluorosulfato)platinate(IV), Cs 2Pt(SO 3F) 6, and Cesium Hexakis(fluorosulfato)antimonate(V), CsSb(SO 3F) 6'. 35 (21): 6113–6130., Schlosberg RH (1968). 'Chemistry in Super Acids. Hydrogen Exchange and Polycondensation of Methane and Alkanes in FSO 3H–SbF 5 ('Magic Acid') Solution. Protonation of Alkanes and the Intermediacy of CH 5 + and Related Hydrocarbon Ions.
The High Chemical Reactivity of 'Paraffins' in Ionic Solution Reactions'. Journal of the American Chemical Society. 90 (10): 2726–7.External links. Titration of acids and simulation of potentiometric titration curves.
